Medical Affairs – Future trends and changing environment
Medical Affairs (MA) focuses to find innovative solutions and continues to evolve fulfilling the needs of all healthcare providers and patients. Following recent trends have emerged in the life science industry.
Patient-Centric Initiatives
- Emphasized patient-centricity that involves engaging the patient population, collaborating, and creating initiatives to satisfy the requirements of the particular patient population.
- Patient’s concerns are prioritized throughout the entire process from initial to post-marketing clinical trial.
- Rebuilds trust between pharmaceutical companies and the patient population.
- Enhanced transparency of the pharmaceutical companies can be brought through the MA team in front of the patients or the subjects.
- Online support forums help patients to interact with their medical experts and listen to their subjects without the fear of being misguided.
Moreover, the life science industries foster patient centricity by:
- Shift from the brand focus to the value of an individual patient
- Engaging patients in research and development (R&D)
- Standardizing the patient-reported outcomes after a thorough understanding of the patients’ experience
- Increasing the patient number
- Convenient medical information
Generation of Real World Evidence
Real-world evidence (RWE) generation is crucial for the MA team to support the patient-centric approach.
- The RWE results in a better insight into the patient journey for the pharmaceutical industry
- Enhances clinical trial diversity: A diverse population ensures the health equity of every patient fulfilling the criteria of meeting the needs of every patient. The U.S. Food and Drug Association (FDA) have guidance for life science organizations to increase the diversity of clinical trials through claims data and electronic health records to maintain a more diverse population.
- Relocation of trial sites close to academic institutions near minority patient communities is another strategy.
Personalized engagement with the MA team
- Life science organizations should provide the MA teams with more personalized unbiased relevant content rather than digital content.
- Effective communication with physicians of content requires proper tailoring. Different specialties need various levels of detail, which must be reflected in the content.
- Each specialty requires specific personalized education. For instance, education initiatives have been outlined to provide special education on biosimilars amongst healthcare professionals. Numerous biosimilar drugs are going to enter the market in 2023 and for many physicians it will be the first time to prescribe. Special emphasis is provided to promote the use of biosimilars amongst healthcare professionals. Moreover, the first board certification has been developed by the Accreditation Council for Medical Affairs for biosimilars and biologics.
All the above trends have one factor in common, a greater focus on the patient and the MA’s journey.
Medical Affairs as the connector breaking down the silo
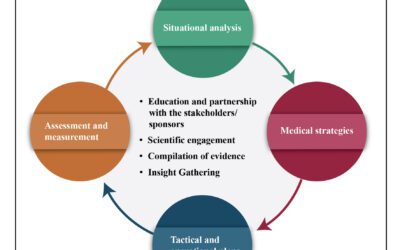
The rapid and continuous flux of pharmacy companies and their frequent adaptation to new protocols provide exciting opportunities but it requires tremendous consistency and alignment challenge for a bigger organization. Hence, this led to a siloed and disconnected organization with extra costs and inefficiencies. A bidirectional flow of information should be encouraged for unbiased medical information from the pharmacy industries to meet the requirements of all stakeholders. MA acts as a bridge in this situation. The role of MA would be to identify the loopholes in the data and be actively involved in modifying strategies accordingly. MAs can also participate in early drug development and identify partnership opportunities.
Involvement of WorkSure® in Medical Affairs
We at WorkSure®, provide customized solutions for MA, medical education and training, medico-marketing and business development, Key Opinion Leader (KOL) management cum interaction, medical communication, literature study, and gap analysis, remodifying the content according to specific requirements.